 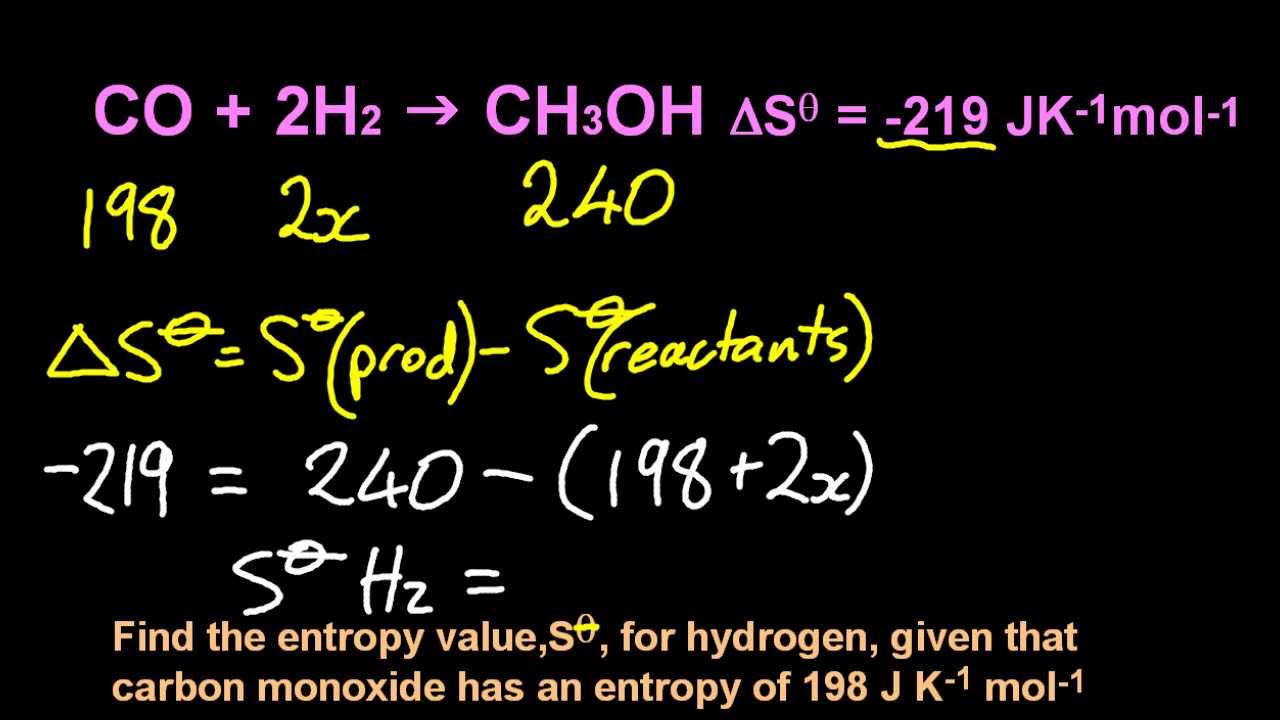
Put the following gases in order of increasing standard molar entropy: CH3CH2OH, He, Ne, and SO2 Lowest Highest B. Gas phase Relative to HBr, reevaluated with current HBr acidity. If the normal boiling point of a liquid is 67 ☌, and. The standard molar entropy at pressure is usually given the symbol S. These are often (but not necessarily) chosen to be the standard temperature and pressure. (a) +6700 J (b) -6700 J (c) +34,000 J (d) -34,000 J. In chemistry, the standard molar entropy is the entropy content of one mole of pure substance at a standard state of pressure and any temperature of interest. 
Gas phase Entropy change calculated or estimated Mīy formula: NO 3 - + H + = HNO 3 Quantity Homework help starts here Science Chemistry If the normal boiling point of a liquid is 67 ☌, and thestandard molar entropy change for the boiling process is +100 J/K, estimate the standard molar enthalpy change forthe boiling process. Reaction search pages in place of the enumerated reaction Go To: Top, Gas phase thermochemistry data, Phase change data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notes Chickos Enthalpy of vaporization Δ vapH (kJ/mol) 
Go To: Top, Gas phase thermochemistry data, Reaction thermochemistry data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notesĭata compiled by: William E. Requires a JavaScript / HTML 5 canvas capable browser. Gas Phase Heat Capacity (Shomate Equation) 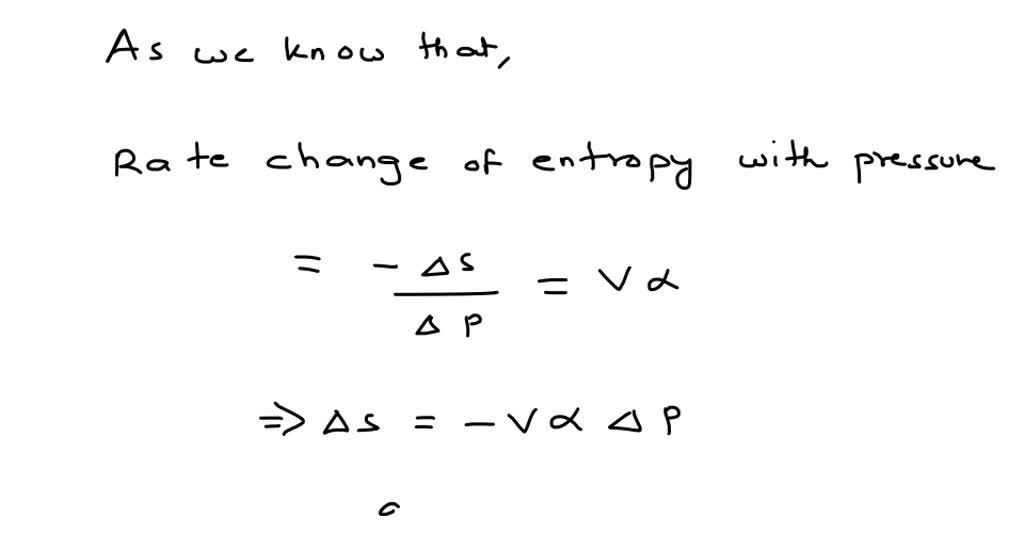
Secretary of Commerce on behalf of the U.S.A. Go To: Top, Phase change data, Reaction thermochemistry data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notesīy the U.S. Your institution may already be a subscriber.įollow the links above to find out more about the data +11.8 J K 1 mol 1 correct incorrect +0. The standard molar constant-pressure heat capacity of hydrogen chloride is 29.1 J K 1 mol 1. With the development of data collections included in Entropy of gas at standard conditions (1 bar) T: Temperature: d(ln(k H))/d(1/T) Temperature dependence parameter for Henry's Law constant: k H: Henry's Law constant at 298.15K: f H gas: Enthalpy of formation of gas at standard conditions: r G Free energy of reaction at standard conditions: r H Enthalpy of reaction at standard. Calculate the change in the standard molar entropy of hydrogen chloride, HCl, gas when it is heated from a temperature of 20☌ to 30☌. The standard entropy of the H+(aq) ion is dened to be 0. These values have been tabulated, and selected substances are listed in Table 18.1a to c Standard Molar Entropies of Selected Substances at 298 K. The purpose of the fee is to recover costs associated 2S g 20.6 205.7 Hg ‘ 0 76.0 Hg+2aq 170.21 32.2 HgO cr,red 90.79 70.3. Standard Molar Entropy, S° The standard molar entropy, S°, is the entropy of 1 mole of a substance in its standard state, at 1 atm of pressure. NIST subscription sites provide data under theĭata Program, but require an annual fee to access.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
